In a clinical trial of PUPs, 42.9% developed inhibitorsb
Designed for the safety and purity you want in your treatment.
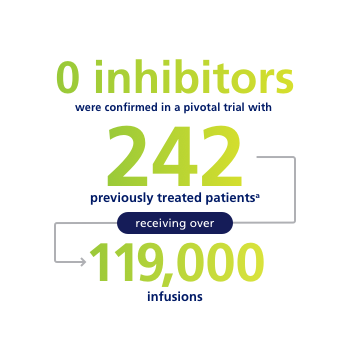
0 inhibitors were confirmed in one of the largest clinical trials programs involving previously treated patients of a rFVIII to date.a
aPeople with previous inhibitors and those new to treatment were not included in the trial. People with hemophilia A may develop inhibitors to factor VIII.
Designed for the safety and purity you want in your treatment.
0 inhibitors were confirmed in one of the largest clinical trials programs involving previously treated patients of a rFVIII to date.a
aPeople with previous inhibitors and those new to treatment were not included in the trial. People with hemophilia A may develop inhibitors to factor VIII.
Sion lives with hemophilia A.
An established safety profile.
- Safety results were consistent among adults, adolescents, and children
- The most frequently reported adverse reactions included swelling or itching at the location of the injection, fever, and inhibitors in previously untreated patients
- Safety results reported after FDA approval have been similar to those observed during clinical trials

In a clinical trial of PUPs, 42.9% developed inhibitorsb
- Safety results were consistent among adults, adolescents, and children
- The most frequently reported adverse reactions in previously treated patients included swelling or itching at the location of the injection, fever, and inhibitors in previously untreated patients
- Safety results reported after FDA approval have been similar to those observed during clinical trials
PUP=previously untreated patient
b59 PUPs with severe hemophilia A (factor VIII level ≤1%) received at least one dose of Novoeight® as part of either routine prophylaxis or on-demand treatment of bleeding episodes. Patients developed inhibitors with a mean of 14.1 exposure days at the time of the first positive inhibitor test. High-risk genetic mutations were identified in 91.7% of the overall inhibitors and 93.3% of the high-titer inhibitors.
Novoeight®
offers purity
through a 5-step
purification
process.


Detergent Inactivation
Helps eliminate enveloped viruses.
Immunoaffinity Chromatography
Uniquely selects intact molecules.

Anion-exchange Chromatography
Separates molecules from impurities based on their charge.

Double Nanofiltration
State-of-the-art double 20-nm filters remove small pathogens, reducing nonenveloped viruses.

Gel Filtration
Reduces FVIII multimers.
Recombinant manufacturing minimizes risk.
Novoeight® is produced by recombinant technology, with no animal- or human-derived components. Recombinant manufacturing minimizes the possibility of viral contamination.
What are the latest recommendations?
The National Hemophilia Foundation’s Medical and Scientific Advisory Council (MASAC) recommends recombinant products as a first-line therapy.

Patient: Rare Bleeding Disorder
MASAC Recommendation: Recombinant
Product if Available
Patient: Previously Treated Patient with Hemophilia A or B
MASAC Recommendation: Recombinant Product
Patient: Previously Untreated Patient with Hemophilia A
MASAC Recommendation: Recombinant FVlll Product or Plasma-Derived VIII Product Containing von Willebrand Factor
What if I’m new to Novoeight®?

It’s easy to get started; we’ll show you step-by-step.
Considering a switch?

It’s important to know how a treatment will fit into your life.
Product support.
Access the product support NovoCare® offers for qualified patients.
