When choosing a hemophilia A treatment, you might also consider extended half-life (EHL) Esperoct® [antihemophilic factor (recombinant), glycopegylated-exei]. We start with the existing Novoeight® molecule and add PEGylation technology to extend the factor’s half-life. The result is up to 50% fewer infusions and factor levels that stay at or above 3% for 100% of the time.b,c
Choosing the treatment that’s right for you.
Whether you’re new to treatment or thinking about making a switch, it’s important to consider all your options. Take a closer look at how the features of Novoeight® may make it the right choice for you:
Choosing the treatment that’s right for you.
Whether you’re new to treatment or thinking about making a switch, it’s important to consider all your options. Take a closer look at how the features of Novoeight® may make it the right choice for you.
David, Dylan and Malcolm live with hemophilia A.
Is it time to reconsider your treatment? Check the standard half-life rFVIII Comparison Guide.a

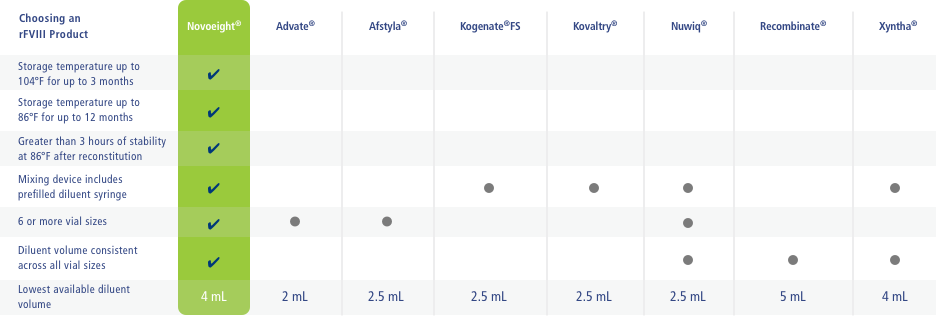
The above table is not intended to compare the safety or efficacy of any of the product.
aData current as of January, 2026.
bTrough level goal is 1% for prophylaxis.
cData shown are from a study where 175 previously treated adolescents and adults received routine prophylaxis with Esperoct® 50 IU/kg every 4 days for 76 weeks. Pre-dose factor activity (trough) levels were evaluated at follow-up visits. Mean trough levels for adolescents (12-<18 years) were 2.7 IU/dL. 50% fewer for adults and adolescents if previously dosed every other day.
Interested in trying Novoeight®?
You may be eligible for our Patient Trial Program. This program provides a free, limited supply of medication for patients new to the treatment. Ask your health care provider to complete the NovoCare® Patient Enrollment Form to get started. Call NovoCare® at 1-844-668-6732 to see if you are eligible.
Portable, reliable, and effective.
Novoeight® is designed to fit into your life. It has the storage flexibility you need to stay on the go, and studies show that it’s reliable.
New to Novoeight®?

It’s easy to get started; we’ll show you step-by-step.
Looking for support?

Novo Nordisk offers educational programs.
